Traces of a animal virus known to infect pigs were found in a 57-year-old Maryland man who survived for two months with a heart transplanted from a genetically altered pig.
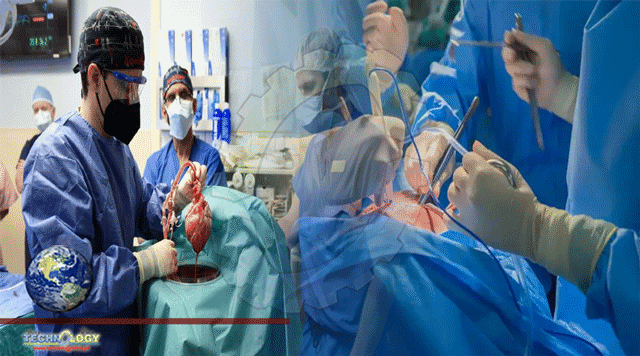
Traces of a virus known to infect pigs were found in a 57-year-old Maryland man who survived for two months with a heart transplanted from a genetically altered pig, according to the surgeon who performed the procedure, the first of its kind.
The disclosure highlights one of the most pressing objections to animal-to-human transplants, which is that widespread use of modified animal organs might facilitate the introduction of new pathogens into the human population.
The presence of the virus’s DNA in the patient may have contributed to his sudden deterioration more than a month after the transplant, said the surgeon, Dr. Bartley Griffith of the University of Maryland School of Medicine.
But there was no evidence that the patient developed an active infection with the virus, or that his body had rejected the heart, Dr. Griffith added.
The patient, David Bennett Sr., had been extremely ill before the surgery and suffered numerous other complications after the transplant. He died on March 8.
Dr. Griffith’s revelations about the viral traces found in the patient, made last month during an American Society of Transplantation meeting, were first reported by MIT Technology Review.
In an interview with The New York Times on Thursday, Dr. Griffith and his colleague, Dr. Muhammad Mohiuddin, the scientific director of the cardiac xenotransplantation program at University of Maryland School of Medical, said that they were saddened by the loss of Mr. Bennett but that they were not deterred from their goal of using animal organs to save human lives.
“This doesn’t really scare us about the future of the field, unless for some reason this one incident is interpreted as a complete failure,” Dr. Griffith said. “It is just a learning point. Knowing it was there, we’ll probably be able to avoid it in future.”
The pig, which had been genetically modified so that its organs would not trigger rejection by the human immune system, was provided by Revivicor, a regenerative medicine company based in Blacksburg, Va.
Company officials declined to comment on Thursday, and officials with the Food and Drug Administration, which gave the transplant surgeons emergency authorization for the operation on New Year’s Eve, said they could not immediately respond to questions.
University officials said that although the pig had been screened several times for the virus, the tests pick up only active infections, not latent ones in which the virus may hide quietly in the pig’s body. (The tests were done on nasal swabs, but the virus was later detected in the pig’s spleen.)
The latent virus might have “hitched a ride” into the patient on the transplanted heart, Dr. Griffith said.
Mr. Bennett’s transplant was initially deemed successful. He did not show signs of rejecting the organ, and the pig’s heart continued to function for well over a month, passing a critical milestone for transplant patients.
A test first indicated the presence of porcine cytomegalovirus DNA in Mr. Bennett 20 days after the transplant, but at such a low level that Dr. Griffith said he thought it might have been a lab error.
About 40 days after the surgery, however, Mr. Bennett suddenly became acutely ill, and subsequent tests showed a precipitous rise in viral DNA levels, Dr. Griffith said.
“So we started thinking that the virus that showed up very early at Day 20 as just a twinkle started to grow in time, and it may have been the actor — it could have been the actor — that set this all off,” Dr. Griffith told other transplant scientists at the meeting.
At Day 45, Mr. Bennett’s health abruptly deteriorated.
Doctors treated Mr. Bennett with antiviral drugs and intravenous immune globulin (IVIG), a product made of antibodies, but the new heart filled with fluid, doubled in size and stopped working, and he was eventually put on a heart-lung machine.
The heart transplant was one of several groundbreaking transplants in recent months that offer hope to the tens of thousands of patients who need new kidneys, hearts and lungs amid a dire shortage of donated human organs.
Surgeons in New York in October successfully attached a kidney grown in a genetically altered pig to a brain-dead patient, and found that the organ worked normally and produced urine.
In January, surgeons at the University of Alabama at Birmingham reported that they had transplanted kidneys from a genetically modified pig into the abdomen of a 57-year-old brain-dead man.
But the prospect of unforeseen consequences — and particularly the potential introduction of animal pathogens into the human population — may dampen enthusiasm for the use of genetically modified organs.
The coronavirus that set off the global Covid pandemic is believed by many scientists to have originated with a virus that was transmitted from an unidentified animal to people in China.
Porcine cytomegalovirus has not been a major concern, since it is a herpes animal virus, which tend to be species-specific, said Dr. Jay Fishman, associate director of the transplantation center at Massachusetts General Hospital, who studies infectious diseases.
“They will replicate only in the host with which they are associated,” Dr. Fishman said.
Nevertheless, the animal virus could infect the transplanted animal organ, leading to a cascade of systemic effects that ultimately harm the patient.
“Did this contribute to the patient’s demise? The answer is obviously, we don’t know, but it might have contributed to his overall not doing well,” Dr. Fishman said.
Dr. Jayme Locke, a transplant surgeon who is director of the Incompatible Kidney Transplant Program at University of Alabama at Birmingham, said genetically modified pigs whose organs are to be used for transplantation must be raised in a pathogen-free facility and weaned from their mothers within 48 hours of birth, in order to prevent transmission of porcine cytomegalovirus during lactation.
The university has such a facility, and Dr. Locke said she was still planning to start a small Phase 1 clinical trial in which she will transplant kidneys from genetically modified pigs into people with end-stage kidney disease.
More sensitive screening of the animals for the virus will be required, she added.
“From my perspective, it’s not slowing down what we need to do, but further emphasizing that data showing our herd is free of that virus will be critical” for regulatory permission to move forward, she said.
This news was originally published by The New York Times.